 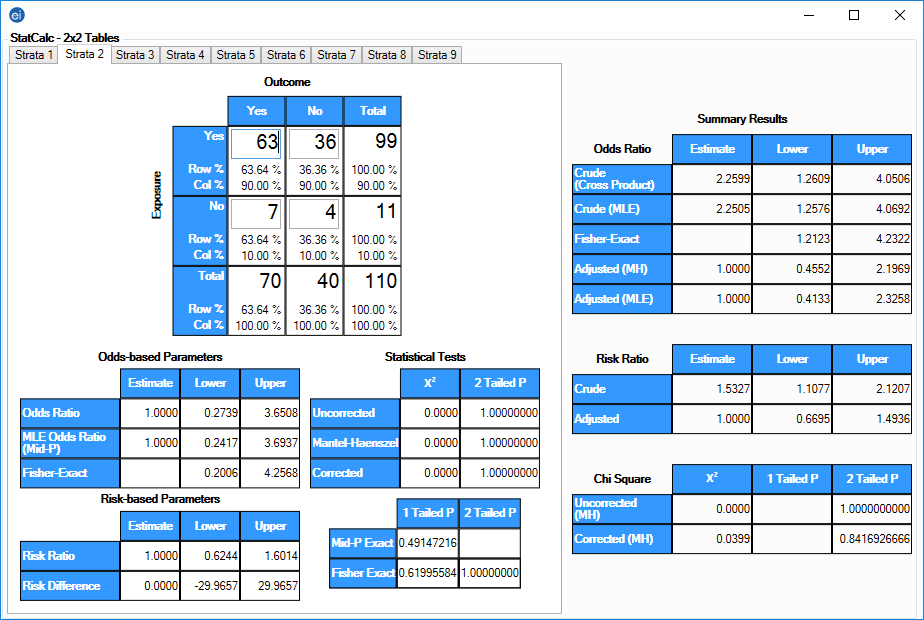
In VD, the calculators can be accessed from the Options menu, under the "Add StatCalc calculator" submenu, and added to the dashboard Canvas in the same manner as other gadgets. The individual calculators are also part of Visual Dashboard (VD). StatCalc appears on the Epi Info™ 7 main menu, middle row, right column, and is captioned "Statistical calculators for sample size, power, and more." Clicking on StatCalc in the main menu opens the StatCalc menu, which is similar to the main menu in appearance and contains buttons for the eight (8) StatCalc calculators listed in StatCalc Calculator Properties, below. The StatCalc tools can be accessed as an independent module from the main menu or as part of the Visual Dashboard. distribution-based event probabilities, 2-tailed p-values and confidence intervals for deviations from binomial (proportions) and Poisson (rare events) distributions given the number of observed and expected events. analysis of 2×2 tables to produce odds ratios and risk ratios (relative risks) with confidence limits, Fisher exact tests, and 1- and 2-tailed p-values, with Mantel-Haenszel summary odds ratios, chi square tests and associated p-values for stratified data 3). sample size and power calculations for unmatched case-control studies, population surveys, cohort or cross-sectional studies, chi-square for trend by the Mantel extension of the Mantel-Haenszel summary odds ratio, and chi square (tests for the presence of a trend in dose-response or other case-control studies where a series of increasing or decreasing exposures is being studied) 2). StatCalc tools can be divided into three broad categories: 1). This journal permits and encourages authors to post and archive the final pdf of the articles submitted to the journal on personal websites or institutional repositories after publication, while providing bibliographic details that credit its publication in this journal.The StatCalc component of Epi Info™ 7 enables the user to evaluate the performance of different study designs and statistical tests by supplying high-level information on the properties of hypothetical data sets and the criteria used for evaluation. In addition, the undersigned affirms that the article described above has not previously been published, in whole or part, is not subject to copyright or other rights except by the author(s), and has not been submitted for publication elsewhere, except as communicated in writing to RHFHSS with this document.Īuthors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License (CC-BY-NC-ND) that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal. The authors hereby transfer, assign, or otherwise convey to RBFHSS: (1) the right to grant permission to republish or reprint the stated material, in whole or in part, without a fee (2) the right to print republish copies for free distribution or sale and (3) the right to republish the stated material in any format (electronic or printed). Of the 65 patients studied, 62 showed potential interactions (95.4%).Ĭonclusion: The high incidence of the use of solid dosage forms and the number of prescription drugs, show that although the available literature, clinical practice is being carried out inconsistently, thus bringing difficulties for the treatment of patients. Twenty-eight (58%) drugs had information on the feasibility of its administration by nasogastric tube. Of the 48 drugs found, 17 (33%) had data in the literature about its possible interactions with nutrition. 
The liquid forms had a low prevalence of use. For most of the drugs were prescribed solid dosage forms. The drugs most prescribed via probe were captopril, lactulose and omeprazole. Were prescribed 3-15 medicines drugs in total and a media of 4 medications per patient by SNE. 
The sample size was calculated through the StatCalc EPI-INFO software version 6.0. The medical record of all patients hospitalized in the clinics II-A and II-B during the period from June to October 2009 and who met the inclusion criteria were analyzed. Methods: This is an observational and descriptive study. 
Objective: To know the characteristics of patients admitted to clinics IIA and IIB using enteral feeding tube (SNE) in a University Hospital Walter Cantídio (HUWC) and the medicines prescribed and used via SNE. Cateter de demora Nutrição enteral Preparações Farmacêuticas Interações Alimento- Abstract 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
